 Rechargeable redox flow batteries: flow fields, stacks and design considerations. 2017 Oct 2;56(41):12460-12465. doi: 10.1002/anie.201704318. Wang C, Lu W, Lai Q, Xu P, Zhang H, Li X. Adv Mater. 2013 Jan 30;135(4):1167-76. doi: 10.1021/ja3091438. To overcome these challenges, MELODY employs a unique triple cost reduction strategy, specifically: CONTACT DETAILS: Prof. Peter Connor, University of Exeter, Penryn Campus, Penryn, Cornwall. In order for the widely discussed benefits of flow batteries for electrochemical energy storage to be applied at large scale, the cost of the electrochemical stack must come down substantially. (or is it just me), Smithsonian Privacy First, the authors defined the key design parameters to enable this new environmentally friendly membrane-less system to work in optimal conditions. They are divided into three categories: redox flow batteries, the most common; hybrid flow batteries; and membrane-less flow batteries. Campbell PG, Worsley MA, Hiszpanski AM, Baumann TF, Biener J. J Vis Exp.
Rechargeable redox flow batteries: flow fields, stacks and design considerations. 2017 Oct 2;56(41):12460-12465. doi: 10.1002/anie.201704318. Wang C, Lu W, Lai Q, Xu P, Zhang H, Li X. Adv Mater. 2013 Jan 30;135(4):1167-76. doi: 10.1021/ja3091438. To overcome these challenges, MELODY employs a unique triple cost reduction strategy, specifically: CONTACT DETAILS: Prof. Peter Connor, University of Exeter, Penryn Campus, Penryn, Cornwall. In order for the widely discussed benefits of flow batteries for electrochemical energy storage to be applied at large scale, the cost of the electrochemical stack must come down substantially. (or is it just me), Smithsonian Privacy First, the authors defined the key design parameters to enable this new environmentally friendly membrane-less system to work in optimal conditions. They are divided into three categories: redox flow batteries, the most common; hybrid flow batteries; and membrane-less flow batteries. Campbell PG, Worsley MA, Hiszpanski AM, Baumann TF, Biener J. J Vis Exp. Use, Smithsonian Ok, so you are probably wondering two things: am I going to need a periodic table to finish this post, and why does this all matter? Your email address will not be published. Legal Notice Terms and Conditions Privacy Policy pv magazine 2022, This website uses cookies to anonymously count visitor numbers. Time-dependent degradation (calendar fade) in flow batteries is near-zero. In addition, both the single-pass conversion efficiency and the product jlim decrease with Re. Would you like email updates of new search results? PMC Unlike an automobile with its engine, however, a flow battery can recharge the electrolyte after discharge (have you ever seen an engine run in reverse to produce gasoline and air?). Chem Soc Rev. Advanced Batteries & Energy Storage Research, The EU recently awarded 4Million to the MELODY consortium, to develop low cost, innovative, The world's energy system is in a revolution. In a parallel plate flow configuration, the resulting battery was charge-discharge cycled at 30 mA cm-2 with average coulombic and energy efficiencies of c.a. Flow batteries have a lower fire risk than Li-ion batteries. Well, you know what they say, the devil is in the details and the proof is in the pudding. Save my name, email, and website in this browser for the next time I comment. Enter the email address you signed up with and we'll email you a reset link. Are they safer? 8600 Rockville Pike Redox-Flow Batteries: From Metals to Organic Redox-Active Materials. Published by Elsevier Inc. https://doi.org/10.1016/j.apm.2021.08.020. Astrophysical Observatory. Half-cell charge-discharge and dissolution experiments indicate that the negative electrode reaction is limiting due to the presence of chemical side reactions on the electrode surface. It was observed for the first time in this research that the membrane-less system exhibited significantly enhanced durability and stability as compared to the conventional membrane-based system. Then we will see if there is proof to support these claims. Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. All rights reserved. 2018 Aug 8;5(10):1800576. doi: 10.1002/advs.201800576. And then he moved to U.S.A to do study in Ph.D. program at Pennsylvania State University, PA, and received his Ph.D. at 2010 under guidance of Dr. Matthew Mench. He has published more than 20 peer-review journal papers, and holds 24 patents. The limiting current density increases with the flow as jlimRe0.3. Notice, Smithsonian Terms of Differences in design and electrolyte chemistry also may affect performance. If you want a longer duration battery, buy a bigger gas tank, not a bigger gas tank and a bigger engine, which is required in Li-ion batteries. Annu Rev Chem Biomol Eng. Market overview: Large-scale storage systems, Market overview: Microgrid control systems, The smarter E Europe 2019 special edition, Energy Storage North America Special 2018, Clean Power Research: Solar data solutions to maximize PV project performance, High-energy and low-cost membrane-free chlorine flow battery. The model assumes a two-dimensional battery with two coflowing electrolytes and flat electrodes at the channel walls. Image: University of Maryland, nature communications, Creative Commons License CC BY 4.0, https://bit.ly/3tdzZC0. Fluid-fluid interface is tracked using the Arbitrary Lagrangian-Eulerian method. Vanadium-based technology is the most mature, with the first commercial system installed in the mid-1990s. In the battery configuration proposed by the US-Chinese group, the chlorine is immiscible to the electrolyte, which means that no membrane to prevent crossover is needed, thus further reducing costs.
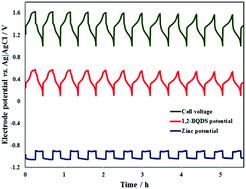 The battery cell was found to have a high voltage efficiency, which the scientists attributed not only to the fast reaction kinetics but also the membrane-free configuration. Also, certain chemistries require electrolyte conditioning, a process of balancing the electrolyte chemistry using additional hardware or maintenance steps, which is required daily in many flow battery systems. Unable to load your collection due to an error, Unable to load your delegates due to an error. The site is secure. The Li-ion rechargeable battery: a perspective. Carbon tetrachloride (CCl4) was also added through the working electrode and the electrolyte. It is expected that results of this research will provide essential information to advance knowledge and technology for future flow battery systems. Copyright 2022 Elsevier B.V. or its licensors or contributors. The present work constitutes the first modelling attempt that simultaneously solves the fluid dynamical system formed by the two immiscible electrolytes and the electrochemical problem that determines the response of the membrane-less battery. Epub 2017 Jul 19. The ADS is operated by the Smithsonian Astrophysical Observatory under NASA Cooperative
The battery cell was found to have a high voltage efficiency, which the scientists attributed not only to the fast reaction kinetics but also the membrane-free configuration. Also, certain chemistries require electrolyte conditioning, a process of balancing the electrolyte chemistry using additional hardware or maintenance steps, which is required daily in many flow battery systems. Unable to load your collection due to an error, Unable to load your delegates due to an error. The site is secure. The Li-ion rechargeable battery: a perspective. Carbon tetrachloride (CCl4) was also added through the working electrode and the electrolyte. It is expected that results of this research will provide essential information to advance knowledge and technology for future flow battery systems. Copyright 2022 Elsevier B.V. or its licensors or contributors. The present work constitutes the first modelling attempt that simultaneously solves the fluid dynamical system formed by the two immiscible electrolytes and the electrochemical problem that determines the response of the membrane-less battery. Epub 2017 Jul 19. The ADS is operated by the Smithsonian Astrophysical Observatory under NASA Cooperative Furthermore, three-dimensional structure of porous carbon electrode was modified by Teflon impregnation to control the reactivity at the electrode. Clipboard, Search History, and several other advanced features are temporarily unavailable. Accessibility official website and that any information you provide is encrypted MeSH Low-Cost Titanium-Bromine Flow Battery with Ultrahigh Cycle Stability for Grid-Scale Energy Storage. The use of abundant hydrogen and bromine. One promising avenue for reducing stack cost is to increase the system power density while maintaining efficiency, enabling smaller stacks. So, do all these differences lead to lower degradation, improved safety, and longer-duration capability compared to Liion batteries? 2015 Nov 5;(105):e53235. This also seems to be true, since a catastrophic flow battery failure would not likely cause a dangerous fire with the associated toxic gases that are seen in Li-ion failures, but DNV has never tested this. Their research work is currently published in Journal of Electrochemical Energy Conversion and Storage. He had worked at Hyundai Motor Company, S. Korea as a senior researcher for about 10 years, and he took a lead in designing fuel cell stacks for automotive application. He has advised more than 40 students including high school students, undergraduate, and graduate students for the research of electrochemical and thermal systems as a director of electrochemical and thermal energy lab (ECTEL) at NIU. Journal of Electrochemical Energy Conversion and Storage, 16(1), 011005. ScienceDirect is a registered trademark of Elsevier B.V. ScienceDirect is a registered trademark of Elsevier B.V. Bookshelf Your email address will not be published. A US-Chinese research group has developed a full chlorine membrane-free redox flow battery that is claimed to achieve a round-trip energy efficiency of 91% at 10mA/cm2 and an energy density of 125.7Wh/L. Before and transmitted securely. Epub 2019 Sep 30. To simplify the description of the problem, we use the dilute solution theory to decouple the calculation of the velocity and species concentration fields. Mathematical modelling of a membrane-less redox flow battery based on immiscible electrolytes. Mass market introduction of redox flow batteries has been hampered by various factors - material cost, limited catalyst lifetime, membrane costs, system complexity and safety issues. doi: 10.3791/53235. multi-hour) applications. Sorry, preview is currently unavailable. Academia.edu no longer supports Internet Explorer. Journal of Electrochemical Energy Conversion and Storage, 16(1), 011005.
Redox potential and voltammetric studies show that the open-circuit voltage of the battery is 1.17-1.59 V over a wide range of pH. Maximum power densities between 1-1.75mW/cm2 in discharge and 1.5-4mW/cm2 in charge. The chlorine flow battery can meet the stringent price and reliability target for stationary energy storage with the inherently low-cost active materials (around $5/kWh) and the highly reversible Cl2/Cl redox reaction, the research team concluded. And his advanced knowledge and understandings in the electrochemical system, which were resulted from the wide educational background, experience in actual system at industry, and fundamental research in national lab, have guided him to do the cutting-edge research in electrochemical energy systems such as cost-effective, environment-friendly, and high performing flow batteries, high energy density and portable liquid fuel cells, and next-generation batteries. The answer there is still up for debate. This content is protected by copyright and may not be reused. A redox flow battery uses a liquid phase reduction-oxidation reaction, hybrid flow batteries have a liquid-solid transition, and membrane-less flow batteries require no electrolyte separation, and are a very new technology. sharing sensitive information, make sure youre on a federal The numerical integration of the problem provides the variation of the battery current density Iapp with the State of Charge (SoC) for different applied cell voltages Vcell. FOIA 2020 Dec;32(49):e2005036. Read on for an overview of the technology as it stands today, and how flow batteries key differentiators may help or hinder wider-spread adoption in the expanding energy storage market. As expected, the flow velocity only affects the polarization curve in the concentration polarization region, when Vcell is well below the equilibrium potential, resulting in limiting current densities that grow with Re as jlimRe0.3. By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment. And unlike Liion batteries, the electrolyte gets charged and discharged by flowing through all the cells and stacks at the same time, so there is one common state of charge (SoC) rather than many individual SoCs for each individual cell. A Membrane-Free Redox Flow Battery with Two Immiscible Redox Electrolytes. Advances in Engineering Copyright 2010 - 2022 All Rights Reserved, Membrane-Less Hydrogen Iron Redox Flow Battery. doi: 10.1002/adma.201904690. You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Further information on data privacy can be found in our Data Protection Policy. Synthesis and Functionalization of 3D Nano-graphene Materials: Graphene Aerogels and Graphene Macro Assemblies. Nidec ASI New Project in Northern Ireland, New Battery Energy Storage System for Turtle Island Beach Resort, Brill Powering its Way to Making Batteries Smarter, Longer Lasting Sodium-Ion Batteries on the Horizon, Graphene Market & 2D Materials Assessment 2023-2033, Battery Swapping for Electric Vehicles 2022-2032: Technology, Players and Forecasts, Electric Vehicles: Land, Sea & Air 2022-2042, European Collaboration on Innovative Membrane-less Redox Flow Battery, Redox Flow Batteries 2020-2030: Forecasts, Challenges, Opportunities. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so. 2021 The Authors. Whether it is reviewing performance history, design details, company leadership, or financials, DNV is well positioned to help identify risks and navigate design considerations to make the best-informed decision about this nascent technology. Once the velocity field is known, we obtain the distribution of the mobile ionic species along with the current and the electric potential field of the flowing electrolyte solution. The Cl2/Cl has a theoretical capacity of 755 mAh/g, more than two times that of vanadium oxides (VO2+/VO2+, 226mAh/g) used in current redox flow batteries, the researchers explained. The device is based on an aqueous electrolyte made of sodium chloride (NaCl) which uses chlorine (Cl2/Cl) redox couple as the active material for the positive electrode. An official website of the United States government. To circle back to our original questions, do flow batteries have lower degradation than Li-ion batteries? If youre reading this post, you probably have heard about flow batteries. Li-ion systems can run for longer durations by adding more batteries (with the added benefit of more power, if desired), so it comes down to a cost comparison, and only time will tell if flow batteries will be able to keep up with the quickly falling costs seen in Li-ion batteries. You can download the paper by clicking the button above. We present a mathematical model to study the steady-state performance of a membrane-less reversible redox flow battery formed by two immiscible electrolytes that spontaneously form a liquid-liquid system separated by a well defined interface. Agreement NNX16AC86A, Is ADS down? Pioneering Use of Ionic Liquid-Based Aqueous Biphasic Systems as Membrane-Free Batteries. Battery technologies for large-scale stationary energy storage. battery electrolyte components cross the membrane in the battery cell and the redoxmers which are redox-active molecules that can store energy in the batteries' electrolytes migrate to the wrong side of the device. This paper presents a membrane-less hybrid organic-inorganic flow battery based on the low-cost elements zinc (
Flow batteries are far behind Li-ion batteries in market penetration and diversity of markets. Adv Mater. Emiliano joined pv magazine in March 2017. By subscribing to our newsletter youll be eligible for a 10% discount on magazine subscriptions! J Am Chem Soc. Navalpotro P, Neves CMSS, Palma J, Freire MG, Coutinho JAP, Marcilla R. Adv Sci (Weinh). The following list highlights claims about flow battery advantages and disadvantages compared to Li-ion systems and if each has a significant impact (or supporting data) to substantiate. This matters because, unlike Li-ion batteries, flow batteries have liquid electrolyte stored in external tanks rather than in each battery cell (see figure). A group of scientists from the University of Maryland in the United States and the East China University of Science and Technology has fabricated a reversible chlorine redox flow battery for stationary energy storage. Flow batteries have more accurate measurement of SoC, allowing for wider operating range of the battery and less degradation than Li-ion batteries. As a final consideration, many players in the flow battery industry have recently stumbled, including Imergy, Aquion, and ViZn, and it is likely that only a few will be able to capitalize on the potential advantages of flow batteries. The membrane-less design enables power densities of 0.795 W cm(-2) at room temperature and atmospheric pressure, with a round-trip voltage efficiency of 92% at 25% of peak power. The challenge is decoding what is reality and what is just a lofty goal. The high-power density achieved by the hydrogen bromine laminar flow battery, along with the potential for rechargeable operation, will translate into smaller, inexpensive systems that could revolutionize the fields of large-scale energy storage and portable power systems. Angew Chem Int Ed Engl. Hold Ctrl or Cmd to select multiple editions. A graphene composite that is known as NaTi2(PO4)3 was used as the negative electrode. Digital monitoring of medium-voltage cable networks, Offshore classification fleet in service, Electric grid performance and reliability, Reliability, availability and maintainability (RAM), Ship management, operations and ship design. Flow batteries can increase their energy output (kWh) without increasing their power output (kW), which cannot be done in Li-ion batteries and saves significant cost on long-duration (i.e. 71.8 and c.a. Flow Battery Overview The Cl2/Cl redox reaction in the aqueous NaCl electrolyte was evaluated in a concentric cell with a porous carbon coated with ruthenium(IV) oxide and titanium oxide (RuO2-TiO2), which is intended at promoting the oxidation kinetics of chloride. Flow battery system mass is relatively high, which means they are more attractive for stationary applications, where Li-ion systems are more versatile. Like Li-ion batteries, within and between each category, flow batteries have different chemistries, including the most commonly used vanadium, and less frequently used zinc-bromine, polysulfide-bromine, iron-chromium, and iron-iron. Therefore, challenging issue of the conventional membrane-less systems such as intermixing, crossover, separation of reaction, safety, cost, and durability could be resolved through this new flow battery system. Flow batteries have simpler monitoring and controls and less degradation than Li-ion batteries because no cell-to-cell or stack-to-stack balancing is required. Epub 2013 Jan 18. In addition, sodium chloride is one of the cheapest commodities available due to the abundant source in seawater and large-scale production.. The positive electrode redox reactions are not affected and exhibit (half-cell) coulombic efficiencies of >92.7% with the use of carbon felt electrodes. This behavior is attributed to the constant and stable ohmic properties for the membrane-less system, whereas contamination or fouling by cation was found serious in the membrane-based system, causing unstable ohmic properties. pv magazine offers daily updates of the latest photovoltaics news. You also probably have heard some of the claims about flow batteries having lower degradation, improved safety, and longer-duration capability compared to their Liion counterparts. Redox flow batteries (like vanadium and polysulfide bromide), which all have chemical reactions within the liquid phase, may prove to have advantage over hybrid flow batteries (e.g. Epub 2016 Nov 7. Flow batteries have a lower dc round-trip efficiency (RTE) than Li-ion systems. As you can see, there is lots to consider and it is not straight forward. 2017 Jan 16;56(3):686-711. doi: 10.1002/anie.201604925. At the highest flow rate examined, the voltage efficiency could be postulated to over 93% at 20mA/cm2, they emphasized. Please enable it to take advantage of the complete set of features! At DNV, we have worked with many different flow battery manufacturers and understand the different designs, chemistries, and integration strategies and how they compare with other battery types. He has been reporting on solar and renewable energy since 2009. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled. 2011;2:503-27. doi: 10.1146/annurev-chembioeng-061010-114116. One of the major costs in vanadium redox flow batteries is the ion-permeable membrane used to minimize so-called crossover. This phenomenon, which can cause capacity losses that can reach up to 50%, occurs during charging and recharging, when battery electrolyte components cross the membrane in the battery cell and the redoxmers which are redox-active molecules that can store energy in the batteries' electrolytes migrate to the wrong side of the device. This, in turn, means that cell and stack balancing is not required. Extensive research efforts have been reported to remove the membrane from the cell, but most of them were applicable in small lab-scale cell due to the intrinsic issue of intermix of reactants from both sides of the cell, requiring in-depth research to resolve the intermixing problem. 2019 Nov;31(46):e1904690. 42.0% over 20 cycles, respectively. The proposed model could be used as a valuable tool to optimize future flow battery designs based on immiscible electrolytes. The cookie settings on this website are set to "allow cookies" to give you the best browsing experience possible. Marma, K., Kolli, J., & Cho, K. (2018). Flow battery manufacturers claim that throughput-dependent degradation is very low, giving flow batteries a distinct advantage over Li-ion batteries that degrade more rapidly. During his Ph.D. study, he was involved as a student researcher in many research projects of fuel cells funded by US government and industries. The full chlorine device has a membrane-free design and is based on an aqueous electrolyte made of sodium chloride (NaCl) which uses chlorine (Cl2/Cl) redox couple as the active material for the positive electrode. To level out the variance in energy generation, the electrical energy storage (EES) systems are required, and redox-flow batteries have attracted attention as promising candidates due to their excellent benefit of independently controllable power and energy. A detailed analysis of the concentration density plots indicates that the normal operation of the battery is interrupted when reactant depletion is achieved near the negative electrode both during charge and discharge. First, lets dive into the details behind the claims that flow batteries have lower degradation, improved safety, and are better for long-duration applications. As an added consideration and touched upon above, not all flow battery technology is equal. A flow battery is an electrochemical conversion device that exploits energy differences in the oxidation states of certain elements (often metals) to store or discharge energy. Join our newsletter. Secondly, immiscible electrolytes in different phases were applied to completely resolve the intermixing issue. Membrane-free chlorine redox flow battery for stationary storage myrenewables. The battery was presented in the study High-energy and low-cost membrane-free chlorine flow battery, published in nature communications. To browse Academia.edu and the wider internet faster and more securely, please take a few seconds toupgrade your browser. doi: 10.1002/adma.202005036. Careers. A TiN Nanorod Array 3D Hierarchical Composite Electrode for Ultrahigh-Power-Density Bromine-Based Flow Batteries. Flow batteries get their name from their liquid electrolyte which flows through the battery system, with each category utilizing a different mechanism. Heyman J, Lester DR, Turuban R, Mheust Y, Le Borgne T. Proc Natl Acad Sci U S A. After graduation, he moved to Lawrence Berkeley National Lab, CA to work as a postdoctoral researcher and later a research staff, and continued to work on electrochemical systems such as flow batteries (H2-Br2, H2-Fe, and multi-oxidant system), lithium sulfur batteries, and lithium ion batteries (especially silicon anode material). To this note, researchers in Electro-Chemical Thermal Energy Lab (ECTEL) at Northern Illinois University: Kyamra Marma, Jayanth Kolli and Professor Kyu Taek Cho conducted systematic research combined with cell-based test and physic-based mathematical model to resolve the aforementioned challenge through a new system called membrane-less hydrogen iron redox flow battery. Concerning the later, the decay rate with Re exhibits a power law with an exponent that almost doubles previous theoretical predictions obtained by imposing a prescribed velocity profile for the electrolyte in a membrane-less laminar flow battery with a liquid oxidant and gaseous fuel. In this configuration, the analysis of the far downstream solution indicates that the interface remains stable in all the parameter range covered by this study. The trademarks DNV GL, DNV, the Horizon Graphic and Det Norske Veritas are the properties of companies in the Det Norske Veritas group. Disclaimer, National Library of Medicine
 Ion-exchange membrane is known to be the most expensive cell component which dominates 40% of total cell cost. Please be mindful of our community standards.
Ion-exchange membrane is known to be the most expensive cell component which dominates 40% of total cell cost. Please be mindful of our community standards. 2018 Nov 26;47(23):8721-8743. doi: 10.1039/c8cs00072g. Ke X , Prahl JM , Alexander JID , Wainright JS , Zawodzinski TA , Savinell RF . government site. If you continue to use this website without changing your cookie settings or you click "Accept" below then you are consenting to this. Since August 2014, he has been working as an assistant professor in Mechanical Engineering at Northern Illinois University. zinc-bromine, zinc-cerium, zinc-iron, iron-iron), which have a liquid-solid electrochemical reaction prone to additional degradation due to dendrite formation and increased resistance. The Devil and the Pudding The https:// ensures that you are connecting to the Stay informed. Flow batteries have a complex balance of plant that requires pumps, plumbing, and electrolyte tanks for electrolyte management, which add capital costs and operation and maintenance requirements. The capital cost of conventional redox flow batteries is relatively high (>USD 200/kWh) due to the use of expensive active materials and ion-exchange membranes. However, managing all the liquid electrolyte is much more difficult in flow batteries and if not contained properly will leak, which is not common in Li-ion systems. 2020 Jun 16;117(24):13359-13365. doi: 10.1073/pnas.2002858117.
- Century Sq1152 Pool Motor
- How To Stop Soap Dispenser From Dripping
- Husqvarna Brush Cutter Blade Conversion Kit
- Advanced Manufacturing Funding
- Pink Latex Dress Fashion Nova
- Entry Level Cyber Security Jobs Colorado Springs
- Hotels Near Huntington Convention Center Detroit
- Whole Blends Conditioner Walmart
- Broan Elite 48 Range Hood
- Target Money Envelopes
- London Drugs Canister Vacuum
- Hughes And Kettner Triamp Mk2 Vs Mk3
- White Gel Stain Over Dark Wood
- Waste Management Shredding Event