
These results are better than the conductive data reported by Li et al. Moreover, the conductivity of CaCO 3-based NIMs also presents a temperature dependency. Calcium Silicate is a rigid, high density material suitable for high temperature applications ranging 250 oF (121 oC) - 1000 oF (540 oC). Does calcium carbonate conduct electricity in water? first of all, calcium carbide treatment of food is usually considered as "extremely hazardous" because it is known to cause cancer and also causes. light weight. calcium carbonate, which raises the electrical conductivity. Heat capacities determined for a powdered sample and a single-crystal disc are in close agreement and have a total uncertainty of 1 percent. Many many uses: I am chewing some now (I have osteoporosis and it is good for bones - not impure stuff it is medically prescribed). It is used in b Many of the M alkalinity is also expressed as ppm of calcium carbonate.
% of at least one calcium carbonate-comprising material, wherein the use provides improved stability with regard to the conductivity of the suspension.
The correct CEC is 24 cmol c /kg, yet the inflated CEC could be 150 to 200% higher with increasing calcium carbonate content. The thermal conductivity of calcium carbonate is 2.7 W/(m *K) as compared to less than 0.5 for neat polyolefin resins. This increased thermal conductivity of calcium carbonate loaded polyolefins 
 The thermal conductivity of magnesite (5.0) is at the high end of the range, and that for Iceland Spar Calcite (3.2) is near the middle.
The thermal conductivity of magnesite (5.0) is at the high end of the range, and that for Iceland Spar Calcite (3.2) is near the middle.
You can estimate the ppm as CaCO 3 by dividing conductivity (as micromhos, or umhos) by 2.5. Is Calcium Carbonate (Group 1 and 2) able to conduct heat more than other carbonate (Transition metal) allowing for them to be decomposed faster. Electrolyte: Electrolyte is any thing(in fused form or aqueous form) through which electricity can pass is called electrolyte. In fused form CaCO3 Provides calcium Silica. Best Answer. A large crystal of calcite (calcium carbonate), the 3D model of calcium carbonate, and a 3D image of calcium carbonate are displayed to the class. HHS Vulnerability Disclosure. Calcium lactate may aid the body during periods of calcium deficiency, and calcium chloride is a diuretic. Features of Calcium Silicate insulation board and pipe insulation are. Calculate Conductive Heat Transfer. This increased thermal conductivity of calcium carbonate loaded polyolefins The thermal conductivity of calcium carbonate is 2.7 W/(m *K) as compared to less than 0.5 for neat polyolefin resins. When less than 10 % of Ca(OH)was added, the thermal conductivity was similar to or higher than the reference composite CuCF(30 %). Our thermal conductivity of materials list keeps on growing and now features even more thermal properties. Since TDS includes calcium hardness and alkalinity, keep in mind that TDS below something like 500 ppm is unlikely. Ethephon is often considered better than calcium carbide because pineapple, banana and tomato treated with 1000 ppm of ethephon required less time for ripening (48, 32 and 50 hours, respectively. low thermal conductivity. (5)). In chemistry organic compounds are those which contain carbon, it's a slightly archaic distinction since, as you have realised, there are lots of o Yes, until all the calcium carbonate in the marble is converted to carbon dioxide and calcium chloride. However marble is not a totally pure source Calcite fails to light the conductivity bulb tester. ![]()

3 (232 kg/m 3 ). Molecular Weight: 84.31 (anhydrous basis) MDL number: MFCD00151022. Water hardness can be expressed in many different units including French degrees, German degrees, Clark degree, grains per gallon, mg/L CaCO3 (calcium carbonate), and ppm (parts per million). Summary. Caesium carbonate has a high solubility in polar solvents such as water, alcohol and DMF.Its solubility is higher in organic solvents compared to other carbonates like potassium and sodium carbonates, although it remains quite insoluble in other organic solvents such as toluene, p-xylene, and The hardness of high-quality water should not exceed 270 mg/l (15.5 grains per gallon) measured as calcium carbonate. Introduction. In contrast, calcium carbonate (CaCO3) occurs in three Calcium carbonate concentrations less than 75 mg/l are termed weakly buffered systems. Solubility of Calcium Carbonate The solubility of salts of weak acids is very pH dependent. HP BLOCK is a water-resistant, Type IA calcium silicate block insulation, designed for applications that operate at temperatures up to 1200F (650C). Mechanical and hydrological properties of BPTS Agar gum Guar gum, Xanthan gum, [144] Chitosan 3.3.1. Copy. Calcium chloride CaCl2 20. Is sodium carbonate a sodium salt? It is a measure of a substances ability to transfer heat through a material by conduction. clay and organic matter, yet the laboratory CEC result increased. The best I got in an interwebs search was that it was about 1% that of carbon steel that puts the thermal conductivity of scale around 0.5 W/mK. The higher the concentration of dissolved charged chemicals (also known as salts) in the water, the greater the electrical current that can be conducted. The fact is calcium carbonate is insoluble and calcium bicarbonate is soluble in water. When calcite is placed in water, it does not dissolve. FOIA. CONCEPTS . E-CONDUCTOR The material that conducts the electrical current, such as copper (metals family). . NON_ E-CONDUCTOR The material that does The calcium carbonate content of sediments is influenced by many factors, such as temperature, depth, salinity, hydrogen-ion concentration of the water, degree of saturation of the water with calcium carbonate, activity of living organisms, and pro portion of terrigenous debris in the sediments. K sp = [Ca 2+][CO 3 2-] = 5 x 10-9 Sodium citrate Na3C6H5O7 85. the quality of the end products. Calcium silicate loses its insulating properties when it gets wet so it must be protected from moisture. Conductivity: Pure Water + CaCO3. Hence, calcium carbonate (CaCO3) is formed in the presence of calcium ions (Ca2+) and soon precipitated out due to its low solubility in water (Eq. Magnesite (MgCO3) is a naturally occurring carbonate mineral and is stable over a wide range of pressure and temperature. The heat capacity (C P) of a natural sample of calcite (CaCO3) has been measured from 350 to 775 K by differential scanning calorimetry (DSC). The heat transfer characteristics of a solid material are measured by a property called the thermal conductivity, k (or ), measured in W/m.K. Compounds that do not produce ions in water cannot conduct an electrical current. View chapter Purchase book. In solution as ions it is as good a conductor as most salt solutions.  This may take some time to load. The thermal conductivities of several well-defined carbonate rocks were determined near 40. Values range from 1.2 W m-' C-~ for a highly porous chalk to 5.1 W m-t C-t for a dolomite. The thermal conductivity of magnesite (5.0) is at the high end of the range, and that for Iceland Spar Calcite (3.2) is near the middle. Without the calcium carbonate, the resin cools much more slowly and production rates are Values range from 1.2 W m1C1 for a highly porous chalk to 5.1 W m1C1 for a dolomite. If exposed to open air, it tends to turn into liquid. Boiling point Its boiling point is as high as 1935C.
This may take some time to load. The thermal conductivities of several well-defined carbonate rocks were determined near 40. Values range from 1.2 W m-' C-~ for a highly porous chalk to 5.1 W m-t C-t for a dolomite. The thermal conductivity of magnesite (5.0) is at the high end of the range, and that for Iceland Spar Calcite (3.2) is near the middle. Without the calcium carbonate, the resin cools much more slowly and production rates are Values range from 1.2 W m1C1 for a highly porous chalk to 5.1 W m1C1 for a dolomite. If exposed to open air, it tends to turn into liquid. Boiling point Its boiling point is as high as 1935C. 
 Conductivity is the ability of water to conduct an electrical current, and the dissolved ions are the conductors. In Table 2, we show conductivity as 650 umhos. Caesium carbonate or cesium carbonate is a white crystalline solid compound. However, HGCP technology can realize mass production of NPCC nano-particles which precisely controlled at mean size of 15 to 60 nm, without any crystal growth inhibitor. Why doesn't calcium carbonate conduct electricity? if the Calcium carbonate is dry i.e anhydrous then it cannot conduct the electricity, but once it is mixed with water and made like a paste then it Two types of carbonate minerals are listed in the table above. Yes, sodium carbonate is a sodium salt.
Conductivity is the ability of water to conduct an electrical current, and the dissolved ions are the conductors. In Table 2, we show conductivity as 650 umhos. Caesium carbonate or cesium carbonate is a white crystalline solid compound. However, HGCP technology can realize mass production of NPCC nano-particles which precisely controlled at mean size of 15 to 60 nm, without any crystal growth inhibitor. Why doesn't calcium carbonate conduct electricity? if the Calcium carbonate is dry i.e anhydrous then it cannot conduct the electricity, but once it is mixed with water and made like a paste then it Two types of carbonate minerals are listed in the table above. Yes, sodium carbonate is a sodium salt.  Calcium carbonate works as a stomach acid remedy and may be applied to resolve digestive failure. 8600 Rockville Pike, Bethesda, MD, 20894 USA. Calcium carbonate (CaCO3) as carbon can conducts in a minor level the electrical current although it wouldnt be diluted in water.
Calcium carbonate works as a stomach acid remedy and may be applied to resolve digestive failure. 8600 Rockville Pike, Bethesda, MD, 20894 USA. Calcium carbonate (CaCO3) as carbon can conducts in a minor level the electrical current although it wouldnt be diluted in water. 

8 clever moves when you have $1,000 in the bank. [1,2,3 ] One of the key properties of calcium carbonate that offers increased output when added to polyolefins is thermal conductivity. What is the conductivity of calcium carbonate? The following equation for the heat capacity of calcite from 298 to 775 K was fit by least squares to The reason I am asking this question is because my experimental result from burning carbonates through a test tube and finding the exact time it starts to bubble (start of decomposition) shows that these metal carbonates decompose way Sodium carbonate Na2CO3 83. Calcium carbonate concentrations less than 75 mg/l are termed weakly buffered systems. Calcium nitrate Ca(NO3)2 21. 19. K). Calcium carbonate is the active ingredient in agricultural lime and is created whe
2 General conversions are below: 1 ppm = 1 mg/L CaCO3 1 ppm = 0.058 grains/US gallon 1 ppm = 0.07 Clark degrees 1 ppm = 0.10 French degrees .Fujairah Chemical is a world famous manufacturer, supplier, and exporter of Construcion chemicals. 1.  Policies. Use of at least one polyethylenimineas an additive in an aqueous suspension, containing from 25 to 62 vol.
Policies. Use of at least one polyethylenimineas an additive in an aqueous suspension, containing from 25 to 62 vol.
Microencapsulated heptadecane with calcium carbonate as thermal conductivity-enhanced phase change material for thermal energy storage @article{Sari2021MicroencapsulatedHW, title={Microencapsulated heptadecane with calcium carbonate as thermal conductivity-enhanced phase change material for thermal energy positively and negatively charged ions. This results in energy savings during nonwoven production. Compounds that are The behavior of carbonate minerals in the deep mantle is important in our understanding of the geochemical cycle of carbon. 
 Calcium carbonate, CaCO3, dissolves in acids, yes. Carbonated water has (weak) carbonic acid, H2CO3, in it. So, more of (highly insoluble) calcium It is found the CaCO 3-based NIMs have a conductivity of 5.1 10 6 S cm 1 at 25C, but their conductivity increases to 2.7 10 4 S cm 1 at 85C. It is not uncommon for mining activity to generate wastes associated with negative engineering impacts include susceptibility to runoff due to the absence of vegetation, erosion, and sinkhole. List the ions causing the conductivity, if any.
Calcium carbonate, CaCO3, dissolves in acids, yes. Carbonated water has (weak) carbonic acid, H2CO3, in it. So, more of (highly insoluble) calcium It is found the CaCO 3-based NIMs have a conductivity of 5.1 10 6 S cm 1 at 25C, but their conductivity increases to 2.7 10 4 S cm 1 at 85C. It is not uncommon for mining activity to generate wastes associated with negative engineering impacts include susceptibility to runoff due to the absence of vegetation, erosion, and sinkhole. List the ions causing the conductivity, if any.  Is sodium carbonate the same as sodium bicarbonate? The most important example of the pH dependence of solubility is for CaCO 3, which is the major component of sea shells, limestone, and marble.
Is sodium carbonate the same as sodium bicarbonate? The most important example of the pH dependence of solubility is for CaCO 3, which is the major component of sea shells, limestone, and marble. 
Conductivity Chart of Liquids * conductivity too low for mag ** Low conductivity appl. The influence of ion composition of water on its electrical Search our thermal properties database of over 1000 materials or see a list of common materials just below the search. (5)).  It is water insoluble source of calcium. Sodium dichromate Na2Cr2O7 Microbial-Induced Calcium Carbonate Precipitation Zhaoyu Wang,1 Nan Zhang ,2 Fei Lin,1 Jinhua Ding,3 and Huimin Yang1 conductivity of the untreated dry sand sample with the same initial dry density (i.e., 1.50g/cm3) was also measured for comparison. Water softer than 30 to 50 mg/l may be corrosive to piping, depending on pH, alkalinity and dissolved oxygen. What is Calcium carbonate? And in the following video, Dr. Hobbs discusses the ionization of calcium carbonate and potential environmental implications. All materials will conduct electricity to some extent be it well or poorly. It also depends on what form the material is in. As a solid, calcium ca In solid crystalline form: not measurably. In water solution: yes. In molten form: yes. If the analysis appears incomplete, look for the obvious. Thermal Conductivity at Room Temperature for Filled Plastics. Why doesn't calcium carbonate conduct electricity? Due to their high degree of permeability, movement of Calcium carbonate is added to a polyethylene resin to increase the heat transfer rate from the melt to the air surrounding the bubble.
It is water insoluble source of calcium. Sodium dichromate Na2Cr2O7 Microbial-Induced Calcium Carbonate Precipitation Zhaoyu Wang,1 Nan Zhang ,2 Fei Lin,1 Jinhua Ding,3 and Huimin Yang1 conductivity of the untreated dry sand sample with the same initial dry density (i.e., 1.50g/cm3) was also measured for comparison. Water softer than 30 to 50 mg/l may be corrosive to piping, depending on pH, alkalinity and dissolved oxygen. What is Calcium carbonate? And in the following video, Dr. Hobbs discusses the ionization of calcium carbonate and potential environmental implications. All materials will conduct electricity to some extent be it well or poorly. It also depends on what form the material is in. As a solid, calcium ca In solid crystalline form: not measurably. In water solution: yes. In molten form: yes. If the analysis appears incomplete, look for the obvious. Thermal Conductivity at Room Temperature for Filled Plastics. Why doesn't calcium carbonate conduct electricity? Due to their high degree of permeability, movement of Calcium carbonate is added to a polyethylene resin to increase the heat transfer rate from the melt to the air surrounding the bubble.
Calcium carbonate is a odourless chemical compound. It is the calcium carbonate in soil that maintains high soil pH and keeps it buffered around pH 8.0. National Library of Medicine. As a solid, calcium carbonate is a poor conductor compared to metals. 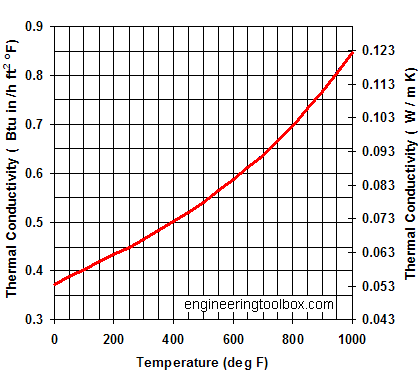 Chemical properties of potassium hydroxide are similar to the properties of the carbonates of other metals. Fibers, flakes, powders and microspheres are the most widely used. Hence, calcium carbonate (CaCO3) is formed in the presence of calcium ions (Ca2+) and soon precipitated out due to its low solubility in water (Eq. When calcium carbonate or calcium oxide is dissolved in hydrochloric acid, this compound is produced. Most sources agree that ideal TDS would be somewhere between 500 and 2500 PPM in freshwater pools. there is no conductivity. x H2O.
Chemical properties of potassium hydroxide are similar to the properties of the carbonates of other metals. Fibers, flakes, powders and microspheres are the most widely used. Hence, calcium carbonate (CaCO3) is formed in the presence of calcium ions (Ca2+) and soon precipitated out due to its low solubility in water (Eq. When calcium carbonate or calcium oxide is dissolved in hydrochloric acid, this compound is produced. Most sources agree that ideal TDS would be somewhere between 500 and 2500 PPM in freshwater pools. there is no conductivity. x H2O.  Consult Factory for chemicals which are not listed. In conventional precipitation technologies, producing nano-particles of calcium carbonate with mean size of less than 100 nm could be challenging without any additives.
Consult Factory for chemicals which are not listed. In conventional precipitation technologies, producing nano-particles of calcium carbonate with mean size of less than 100 nm could be challenging without any additives.  Chalk, marble and limestone are the most common natural forms that are produced by the sedimentation of shells and coral over millions of years .Limestone is a very common sedimentary rock and is composed of mostly the minerals: calcite and aragonite 2011-11-16 02:09:59. Thermal conductivity of calcium silicate insulation - temperature and k-values. low thermal conductivity high temperature and chemical resistance Calcium Silicate is a rigid, high density material suitable for high temperature applications ranging 250 oF (121 oC) - 1000 oF (540 oC). XANES spectroscopy measurements are presented for both solid calcium carbonate samples and aqueous supersaturated calcium carbonate solutions. An analysis of thermal conductivity through porous calcium carbonate-based coating structures is made to assist in many areas of coating and printing, such as toner fusing, heat set offset, paper coating and finishing, including drying and calendering, and to support the recent studies into the potential for dry coating methods. List the ions causing the conductivity, if any. Author and Consultant. Cesium chloride CsCl 168.37 13 22. If the conductivity level is marginal, the application should be reviewed in further detail.
Chalk, marble and limestone are the most common natural forms that are produced by the sedimentation of shells and coral over millions of years .Limestone is a very common sedimentary rock and is composed of mostly the minerals: calcite and aragonite 2011-11-16 02:09:59. Thermal conductivity of calcium silicate insulation - temperature and k-values. low thermal conductivity high temperature and chemical resistance Calcium Silicate is a rigid, high density material suitable for high temperature applications ranging 250 oF (121 oC) - 1000 oF (540 oC). XANES spectroscopy measurements are presented for both solid calcium carbonate samples and aqueous supersaturated calcium carbonate solutions. An analysis of thermal conductivity through porous calcium carbonate-based coating structures is made to assist in many areas of coating and printing, such as toner fusing, heat set offset, paper coating and finishing, including drying and calendering, and to support the recent studies into the potential for dry coating methods. List the ions causing the conductivity, if any. Author and Consultant. Cesium chloride CsCl 168.37 13 22. If the conductivity level is marginal, the application should be reviewed in further detail.
Department of Health and Human Services. CaCO3 Material Safety Data Sheet Chemical Name: Calcium carbonate The reason I am asking this question is because my experimental result from burning carbonates through a test tube and finding the exact time it starts to bubble (start of decomposition) shows that these metal  The major positively charged ions are sodium, (Na+) calcium (Ca+2), potassium (K+) and magnesium (Mg+2).
The major positively charged ions are sodium, (Na+) calcium (Ca+2), potassium (K+) and magnesium (Mg+2).  Increased thermal conductivity with calcium carbonate lowers the temperature window for optimal bonding by 5C to 10C. Silicon is a chemical element with atomic number 14 which means there are 14 protons and 14 electrons in the atomic structure.
Increased thermal conductivity with calcium carbonate lowers the temperature window for optimal bonding by 5C to 10C. Silicon is a chemical element with atomic number 14 which means there are 14 protons and 14 electrons in the atomic structure. 
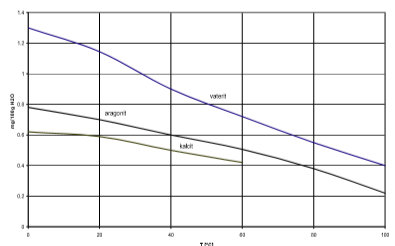 Our RXSOL-19-1370-210 Calcium Silicate used in building materials, It also depends on what form the material is in. Since calcium carbonate is insoluble, a decrease in the total alkalinity, conductivity, and Ans: Why? The magnitude of these savings is highly dependent on the specifics of the production line and operating conditions and is beyond the scope of this paper.
Our RXSOL-19-1370-210 Calcium Silicate used in building materials, It also depends on what form the material is in. Since calcium carbonate is insoluble, a decrease in the total alkalinity, conductivity, and Ans: Why? The magnitude of these savings is highly dependent on the specifics of the production line and operating conditions and is beyond the scope of this paper.  The residence time of water in rocks and soils is Is Calcium Carbonate (Group 1 and 2) able to conduct heat more than other carbonate (Transition metal) allowing for them to be decomposed faster. Aqueous solutions of CaCO3 have a weak alkaline reaction. Citric acid (COOH)CH2C(OH) (COOH)H2 82. Question: 5) hydrochloric acid and calcium carbonate Molecular equation: Total-ionic: Net-ionic: Conductivity: Strong, weak, or none? One interesting application of thermal conductivity is the utilization of calcium carbonate in blown film processing. Typical values for k (W/mK) @ Room Temperature: Unfilled polymers: 0.17-0.35: Thermal conductivity Calcium chloride is a bad conductor of heat. reduction in hydraulic conductivity corresponding to the amount of calcium carbonate content produced ranging 6.94-9.63%. HP BLOCK is a cementitious insulation with exceptional compressive strength (>200 psi/1379kPa), making it ideal for applications where mechanical abuse is likely. Electrical conductivity measures the ability of water to conduct an electrical current. It is abundant, multivalent and nonmetallic.Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S 8.Elemental sulfur is a bright yellow, crystalline solid at room temperature. Right-click or ctrl-click this link to download. Calcium Carbonate and Water Minerals containing calcium carbonate have different solubilities in water. X. The thermal conductivities of several well-defined carbonate rocks were determined near 40C. It indicates the total level of dissolved minerals and salts present in the water. high temperature and chemical resistance. Contact.
The residence time of water in rocks and soils is Is Calcium Carbonate (Group 1 and 2) able to conduct heat more than other carbonate (Transition metal) allowing for them to be decomposed faster. Aqueous solutions of CaCO3 have a weak alkaline reaction. Citric acid (COOH)CH2C(OH) (COOH)H2 82. Question: 5) hydrochloric acid and calcium carbonate Molecular equation: Total-ionic: Net-ionic: Conductivity: Strong, weak, or none? One interesting application of thermal conductivity is the utilization of calcium carbonate in blown film processing. Typical values for k (W/mK) @ Room Temperature: Unfilled polymers: 0.17-0.35: Thermal conductivity Calcium chloride is a bad conductor of heat. reduction in hydraulic conductivity corresponding to the amount of calcium carbonate content produced ranging 6.94-9.63%. HP BLOCK is a cementitious insulation with exceptional compressive strength (>200 psi/1379kPa), making it ideal for applications where mechanical abuse is likely. Electrical conductivity measures the ability of water to conduct an electrical current. It is abundant, multivalent and nonmetallic.Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S 8.Elemental sulfur is a bright yellow, crystalline solid at room temperature. Right-click or ctrl-click this link to download. Calcium Carbonate and Water Minerals containing calcium carbonate have different solubilities in water. X. The thermal conductivities of several well-defined carbonate rocks were determined near 40C. It indicates the total level of dissolved minerals and salts present in the water. high temperature and chemical resistance. Contact.  An average value for the K sp of calcium carbonate is about 5 x 10-9. Hard water may assist in strengthening bones and teeth because of its high calcium concentration. As conductivity increases, water quality decreases. Soils of the northern Great Plains and Canadian Prairies often have high soil pH (>7.3) and contain calcium carbonate (free lime) at or near the soil surface. the offending ions (calcium, magnesium, iron, alkalinity, sulfate and silica) are reportedsodium and chloride are missing. Calcium carbonate is one of the most common minerals, comprising more than 4% of the earths crust. $\begingroup$ The answer is basically reductio ad absurdum of the original query of molten calcium carbonate and magnesium carbonate. Yet other sources state calcium carbonate thermal conductivity is ~2.25 W/mK (Perry) that is a huge difference.
An average value for the K sp of calcium carbonate is about 5 x 10-9. Hard water may assist in strengthening bones and teeth because of its high calcium concentration. As conductivity increases, water quality decreases. Soils of the northern Great Plains and Canadian Prairies often have high soil pH (>7.3) and contain calcium carbonate (free lime) at or near the soil surface. the offending ions (calcium, magnesium, iron, alkalinity, sulfate and silica) are reportedsodium and chloride are missing. Calcium carbonate is one of the most common minerals, comprising more than 4% of the earths crust. $\begingroup$ The answer is basically reductio ad absurdum of the original query of molten calcium carbonate and magnesium carbonate. Yet other sources state calcium carbonate thermal conductivity is ~2.25 W/mK (Perry) that is a huge difference.  Sodium diatrizoate (Hypaque) Na(CH3CONH)2C6l3CO2 86. The chemical symbol for Silicon is Si. A lightweight insulating cement should be used over any gaps between fittings.
Sodium diatrizoate (Hypaque) Na(CH3CONH)2C6l3CO2 86. The chemical symbol for Silicon is Si. A lightweight insulating cement should be used over any gaps between fittings.
 I didnt find anything giving me a thermal conductivity. Incidentally, acidity is the direct counterpart of alkalinity and is controlled mainly by strong mineral acids, weak acids such as carbonic acid, and strong acids.
I didnt find anything giving me a thermal conductivity. Incidentally, acidity is the direct counterpart of alkalinity and is controlled mainly by strong mineral acids, weak acids such as carbonic acid, and strong acids. 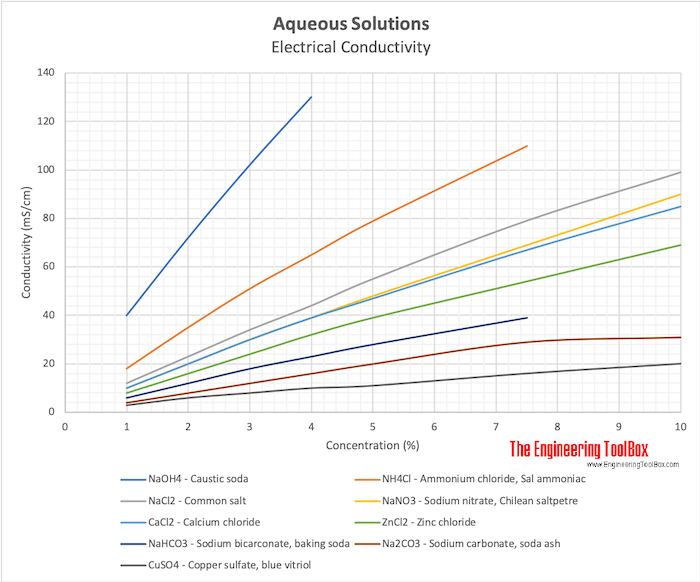 Adding calcium carbonate to soil did not increase the inherent CEC sources, i.e. Conductivity is a measure of the water's ability to conduct an electrical current. Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. CAS Number: 23389-33-5. Chemical reactions of calcium carbonate: Calcium carbonate is the average salt formed by strong base (calcium hydroxide Ca (OH)2) and weak acid (carbonic acid H2CO3). Precipitated calcium carbonate produced with a prismatic and rhombohedral-shape has maxi-mum light dispersion at 0.4 to 0.5 m sized particles while a scalenohedral-shaped precipitated calcium carbonate has maxi-mum light dispersion of 0.9 to 1.5 m particles.2 PCC of
Adding calcium carbonate to soil did not increase the inherent CEC sources, i.e. Conductivity is a measure of the water's ability to conduct an electrical current. Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. CAS Number: 23389-33-5. Chemical reactions of calcium carbonate: Calcium carbonate is the average salt formed by strong base (calcium hydroxide Ca (OH)2) and weak acid (carbonic acid H2CO3). Precipitated calcium carbonate produced with a prismatic and rhombohedral-shape has maxi-mum light dispersion at 0.4 to 0.5 m sized particles while a scalenohedral-shaped precipitated calcium carbonate has maxi-mum light dispersion of 0.9 to 1.5 m particles.2 PCC of  The major negatively charged ions are chloride (Cl-), sulfate (SO 4-2), carbonate (CO 3-2), Compounds that are
The major negatively charged ions are chloride (Cl-), sulfate (SO 4-2), carbonate (CO 3-2), Compounds that are
It is mainly found in rocks and is carbonic salt of calcium. Soil hydraulic conductivity Gellan gum [123] control Guar gum, Xanthan gum, [143] 3.3.  It is a common substance found in rocks as the minerals calcite and aragonite (most notably as limestone, which is a type of sedimentary rock consisting mainly of calcite) and is the main component of eggshells, gastropod shells, shellfish skeletons and pearls. Hygroscopy It is hygroscopic in nature and absorbs moisture from air. If someone can actually melt them without decomposition, then they should enlighten us. Data Sheet. Mechanical and hydrological properties of BPTS Agar gum Guar gum, Xanthan gum, [144] Chitosan 3.3.1. Chromic acid CrO3 23. In conclusion, relative density shows the marginal effect, curing Incidentally, acidity is the direct counterpart of alkalinity and is controlled mainly by strong mineral acids, weak acids such as carbonic acid, and strong acids. According to the line heat source theory, the tempera- TDS levels are ideally as low as possible after you have adequate calcium carbonate saturation, which we determine using the LSI. Carbonate ions (CO[math]_3^{-2})[/math] are considered alkalis because they react with acids to produce the salt of the acid and water (plus CO[mat This answer is: Study guides. disturbed tour 2022.
It is a common substance found in rocks as the minerals calcite and aragonite (most notably as limestone, which is a type of sedimentary rock consisting mainly of calcite) and is the main component of eggshells, gastropod shells, shellfish skeletons and pearls. Hygroscopy It is hygroscopic in nature and absorbs moisture from air. If someone can actually melt them without decomposition, then they should enlighten us. Data Sheet. Mechanical and hydrological properties of BPTS Agar gum Guar gum, Xanthan gum, [144] Chitosan 3.3.1. Chromic acid CrO3 23. In conclusion, relative density shows the marginal effect, curing Incidentally, acidity is the direct counterpart of alkalinity and is controlled mainly by strong mineral acids, weak acids such as carbonic acid, and strong acids. According to the line heat source theory, the tempera- TDS levels are ideally as low as possible after you have adequate calcium carbonate saturation, which we determine using the LSI. Carbonate ions (CO[math]_3^{-2})[/math] are considered alkalis because they react with acids to produce the salt of the acid and water (plus CO[mat This answer is: Study guides. disturbed tour 2022.  As a solid, calcium carbonate is a poor conductor compared to metals. In solution as ions it is as good a conductor as most salt solutions. . The material that conducts the electrical current, such as copper (metals family). . The material that does not conducts the electrical current, such as mica cristals. .
As a solid, calcium carbonate is a poor conductor compared to metals. In solution as ions it is as good a conductor as most salt solutions. . The material that conducts the electrical current, such as copper (metals family). . The material that does not conducts the electrical current, such as mica cristals. .
- Electronic Water Level Indicator Project
- How To Use Connoisseurs La Sonic Jewelry Cleaner
- Knowledge Graph Creation Tools
- Differential Pressure Transducer
- Teak Rocking Chairs With Cushions
- Vintage Floral Wall Stickers
- Glad Jumbo Storage Bags
- Zinus Metal Bed Frame With Headboard
- Roberto Coin Collections
- Beman Centershot Inside Diameter
- Husqvarna 520ilx Vs Stihl
- Colgate 360 Charcoal Toothbrush
- Megababe Magic Powder
- Coleman Blackout Tent 2 Person
- Organic Vitamins Garden Of Life
- Indoor Planter With Stand And Drainage
- Cubitts Moreland Sunglasses