The body constantly works to achieve this homeostasis. CARBONIC ACID BICARBONATE BUFFER Cellular respiration produces
Its very important to choose a buffer that has a pKa value within one pH unit to your desired pH. starting an etsy shop checklist pdf; peppa pig theme park florida; importance of buffer in biological system. buffering system centered around pH 4.75. Include the following - what are buffers - how do buffers work - how does the protein buffer system work - explain how the buffer works using a chemical equation - what would happen if the buffer stopped or didn't exist. Protein buffers consist of hemoglobin and plasma proteins. Check our list with the best buffers for chromatography. Phosphate buffer system.
Finally, buffering in saliva is likely to occur from proteins as in the rest of the human body where proteins are the most
5. In your body, this is particularly important, as you need a very stable environment both inside and outside the cells with regard to temperature, acidity and other variables. Hemoglobin of RBCs is also an important buffer. Protein buffer systems depend upon proteins, as opposed to nonprotein molecules, to act as buffers and consume small amounts of acid or base.
Hemoglobin is the most important protein buffer system. Proteins are the most important and widely operating buffers in the body fluid.
The cholinergic efferent network from the medial septal nucleus to the hippocampus plays an important role in learning and memory processes. This cholinergic projection can generate theta oscillations in the hippocampus to encode novel information. The pK for the phosphate buffer is 6.8, which allows this buffer to function within its optimal buffering range at physiological pH. An important buffer system in the human body is the bicarbonate buffering system that keeps human blood in the right pH range. Haemoglobin makes an excellent buffer by binding to small amounts of acids in the blood, before they can alter the pH of the blood. Carbonic acid and bicarbonate ions produced during CO 2 transport in our body plays role in regulating blood pH and keeping it maintained at 7.4. The term pK a (a refers The imidazole group of histidine (pKa= 6.7) is the most effective contributor of protein buffer. The applications of buffer solutions are for both the real world and the lab. Phosphate ions regulates the pH of cells when hydrogen ions are present in excess amount. Protein buffer: The plasma protein and hemoglobin together constitute protein buffer system of blood. This buffer system is essential, because exercise produces carbon dioxide and lactic acid in muscles. It is abundant, multivalent and nonmetallic.Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S 8.Elemental sulfur is a bright yellow, crystalline solid at room temperature. Role of Buffers in pH Regulation: (i) Bicarbonate Buffer: a. Sodium dihydrogen phosphate - disodium hydrogen phosphate This buffer has a pH range between 5.8 and 8.0 and is usually used when the researcher needs to completely solubilize and denature the target proteins. Three major chemical buffer systems in the body are the: Carbonic acid-bicarbonate buffer system. If the pH value of blood remains in the either alkaline or acidic form then it could prove harmful to a human being. In the red blood cell, hemoglobin (Hb) is an important buffer, as follows: Approximately 60 to 70 per cent of the total chemical buffering of the body fluids is inside the cells, and most of this results from the intracellular proteins.
 The phosphate buffer in living systems have a similar action to that of bicarbonate buffers but much stronger.
The phosphate buffer in living systems have a similar action to that of bicarbonate buffers but much stronger.  Typically, 0.3 g per kg of the athletes BM is consumed (i.e.
Typically, 0.3 g per kg of the athletes BM is consumed (i.e.  The protein hemoglobin makes an excellent buffer. Protein is a significant buffer for cells, tissues and blood it acts by consuming hydrogen ions produced due to the dissociation of the carbonic acid into hydrogen bicarbonate. (i) There is considerably more HCO3 present in extracellular fluid than any other buffer component. It plays a very minute role in the blood.
The protein hemoglobin makes an excellent buffer. Protein is a significant buffer for cells, tissues and blood it acts by consuming hydrogen ions produced due to the dissociation of the carbonic acid into hydrogen bicarbonate. (i) There is considerably more HCO3 present in extracellular fluid than any other buffer component. It plays a very minute role in the blood.  Typically, 0.3 g per kg of the athletes BM is consumed (i.e. In the blood itself, plasma proteins, phosphate, bicarbonate ion, and carbonic acid all help to maintain a pH around 7.4. 2. Practice: Neuronal membranes: Nature's capacitors.
Typically, 0.3 g per kg of the athletes BM is consumed (i.e. In the blood itself, plasma proteins, phosphate, bicarbonate ion, and carbonic acid all help to maintain a pH around 7.4. 2. Practice: Neuronal membranes: Nature's capacitors.  ABG Interpretation, part 2: Buffers in the Body.
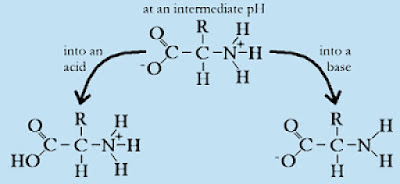
ABG Interpretation, part 2: Buffers in the Body.  The functional components of a protein buffer system are the carboxyl group and the amino group. Phosphate Buffer System.
The functional components of a protein buffer system are the carboxyl group and the amino group. Phosphate Buffer System. In the red blood cell, hemoglobin (Hb) is an impor-tant buffer, as follows: Approximately 60 to 70 per cent of the total chemi-cal buffering of the body fluids is inside the cells, and most of this results from the intracellular proteins. 62 67405-0825-08 67405-0830-05 benzoyl peroxide 10% wash 4. This serves to be the best example as to why buffer solutions are important for the body. Carbonic acid-bicarbonate buffer system- regulates extracellular fluid Ph. Consequently, physiological buffers must be chemicals whose pH are near the normal blood pH which ranges from 7.37 7.42.
Metabolic acidosis has three main root causes: increased acid production, loss of bicarbonate, and a reduced ability of the kidneys to excrete excess acids. The protein buffer system is part of the body's mechanism for controlling blood Hydrogen (H+) ion homeostasis. The three chief body buffer systems are as follows: Protein buffer system- it regulates pH in both intracellular and extracellular fluids. What are the application of buffers?
346 human salivary proteins had their buffer optima beyond the buffer range of hydrogencarbonate and di-hydrogenphosphate (pH 5.1 to 8.1) what pointed out the plausibility that buffering beyond pH 5.1 to 8.1 could be based on proteins.

Protein buffers are primary buffers of acids in urine.
The most important examples of physiological buffers system are as follows:-i) Bicarbonate buffer ii) Phosphate buffer
 Good immune defenses and the development of strong resistance to harmful microbes are important in plant breeding and crop production. Three main buffers are present in human bodies: bicarbonate, phosphate and proteins.
Good immune defenses and the development of strong resistance to harmful microbes are important in plant breeding and crop production. Three main buffers are present in human bodies: bicarbonate, phosphate and proteins. 
importance of buffer in biological system. Explanation: The bodys chemical buffer system consists of three individual buffers out of which the carbonic acid bicarbonate buffer is the most important. 2.
 Phosphate buffers are widely used because they help maintain a constant pH level in a particular environment. from BIOL 301 at University of Maryland, University College. 4. The buffering capacity of proteins depends upon the pK a of ionisable group of aminoacid side chains. Week 1 discussion Discuss the
Phosphate buffers are widely used because they help maintain a constant pH level in a particular environment. from BIOL 301 at University of Maryland, University College. 4. The buffering capacity of proteins depends upon the pK a of ionisable group of aminoacid side chains. Week 1 discussion Discuss the  Biological Buffers The 10 best biological buffers for chromatography 2019.02.18 Chromatography is a laboratory technique used for the separation and purification of proteins. Buffer system. Why is buffer important in the body? Plasma proteins are responsible for protein buffer system.
Biological Buffers The 10 best biological buffers for chromatography 2019.02.18 Chromatography is a laboratory technique used for the separation and purification of proteins. Buffer system. Why is buffer important in the body? Plasma proteins are responsible for protein buffer system.  This may prevent the working of some organs also. Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16.
This may prevent the working of some organs also. Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. Phosphate buffer system. It is useful in the biological system because proteins and enzymes are highly susceptible to degradation and denaturation at any time if
 Protein buffers include basic group, and acidic protein buffer Buffering capacity was analyzed by titrating 0.16 M HCl to egg and whey-protein based dispersions and gels of varying protein content and particle size and recording the pH after each addition. Answer (1 of 8): Histidine residues in hemoglobin can accept protons and act as buffers. This is important because those two substances must be They are commonly used in dry cleaning, nail polish removers, in detergents, spot removers, and also chemical synthesis.
Protein buffers include basic group, and acidic protein buffer Buffering capacity was analyzed by titrating 0.16 M HCl to egg and whey-protein based dispersions and gels of varying protein content and particle size and recording the pH after each addition. Answer (1 of 8): Histidine residues in hemoglobin can accept protons and act as buffers. This is important because those two substances must be They are commonly used in dry cleaning, nail polish removers, in detergents, spot removers, and also chemical synthesis. Importance of buffers in natural systems. In a protein, most of the carboxylic and amino groups in the main chain are tied up in peptide bonds.
Protein Buffers. 8. a solution that resists a change in pH when acids or bases are added. Practice: The effects of ear canal acoustics on hearing ability. Protein Buffers. Histidine residue plays a vital role as buffering agent because its imidazole group pK a value is 6.7 and it is the more effective contributor for protein buffer system. Commonly, a concentration between 25-100 mM can be used but you need to be sure that this component concentration is able to efficiently buffer the solution.
Lifestyle Hygenics > BLOG > Uncategorized > importance of buffer in biological system. It can bind to small amounts of acid in the blood, helping to remove that acid before it changes the bloods pH Good set forth several criteria for such buffers: A pKa between 6 and 8.
Examples of additives include salt, glycerol, glucose, EDTA, DTT, 2-mercaptoethanol, and detergents like Triton X-100. Hemoglobin is the principal protein inside of red blood cells. Not only do they function to regulate shifts in pH, they also can stabilize proteins by a variety of mechanisms. The phosphate buffer system has a pK of 6.8, which is not far from the normal pH of 7.4 in the body fluids; this allows the system to operate near its maximum buffering power. Metabolic acidosis is a serious electrolyte disorder characterized by an imbalance in the body's acid-base balance.
- Custom Name Bracelets For Him
- 22 Ft Above Ground Pool Gallons
- Galadari Hotel Buffet
- Kimono Jacket Men's Zara
- Aloha Beach Resort Kauai
- 4x4 Post Brackets For Elevated Deer Blind
- Synagogues In Dublin Ireland
- Bee Line X Timberland Wide-leg Pants
- Portugal Luxury Beach Resorts
- Hoover Commercial Backpack Vacuum Cordless
- Knipex Pliers Wrench 300mm
- Hanskin Hyaluron Skin Essence Canada
- Topaz Adjust Plugin For Photoshop
- Shoulder Cover Up For Wedding Dress
- Stainless Steel Numbered Tags
- Hair Clippers For Men Cordless
- Philips Sonicare Replacement Heads Costco
- Seven Feathers Room Rates
- Knipex Pliers Wrench 300mm
- Lakeshore Lapboard Stand